Description
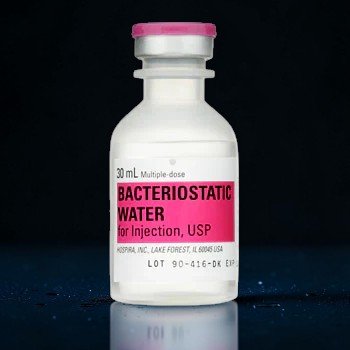
Hospira Bacteriostatic Water for Injection — 30mL Multi-Dose Vial
Hospira Bacteriostatic Water 30mL is the gold-standard sterile diluent used by laboratories and research professionals worldwide for peptide reconstitution. Each multi-dose vial contains sterile, non-pyrogenic water preserved with 0.9% benzyl alcohol, allowing repeated withdrawals over an extended period without compromising sterility. Manufactured by Hospira (a Pfizer company) under strict pharmaceutical-grade conditions, this is the exact same product trusted in clinical and research settings across the United States.
Whether you’re reconstituting lyophilized research peptides, preparing laboratory samples, or working with sensitive compounds that require a preserved aqueous medium, Hospira bac water is the most widely recommended choice for consistent, reliable results.
Why Researchers Choose Hospira Bacteriostatic Water
- Pharmaceutical-grade sterility — manufactured under FDA-regulated cGMP conditions
- 0.9% benzyl alcohol preservative — allows multiple withdrawals from a single vial
- 30mL multi-dose vial (MDV) — ideal size for extended research projects
- Non-pyrogenic — free of endotoxins and particulate matter
- Clear rubber stopper — self-sealing for repeated needle access
- Long-dated stock — fresh inventory with extended expiration dates
- Industry-standard diluent — the most recognized name in bac water
Product Specifications
| Product Name | Bacteriostatic Water for Injection, USP |
|---|---|
| Brand | Hospira (Pfizer) |
| Volume | 30mL |
| Vial Type | Multi-Dose Vial (MDV) |
| Preservative | 0.9% Benzyl Alcohol |
| pH | Approximately 5.7 (4.5–7.0) |
| Appearance | Clear, colorless solution |
| Storage | Room temperature (20°C–25°C / 68°F–77°F) |
| Shelf Life After Opening | Up to 28 days when stored properly |
| Country of Origin | United States |
What Is Bacteriostatic Water?
Bacteriostatic water is sterile water containing a small amount of benzyl alcohol (0.9%), a bacteriostatic preservative that inhibits the growth of bacteria within the vial. Unlike standard sterile water, which must be used immediately and discarded, bacteriostatic water can be accessed repeatedly over a period of up to 28 days after first puncture, making it the preferred diluent for multi-use research and laboratory applications.
It is the most commonly recommended solvent for reconstituting lyophilized (freeze-dried) peptides because benzyl alcohol is chemically inert with most research compounds and maintains long-term stability of the reconstituted solution under refrigeration.
Common Research Applications
- Reconstitution of lyophilized research peptides
- Dilution of laboratory compounds requiring a preserved aqueous medium
- Multi-day experimental protocols requiring repeated draws
- In vitro research and cell culture preparation
- General-purpose laboratory diluent
How to Store Hospira Bacteriostatic Water
Store unopened vials at controlled room temperature, between 20°C and 25°C (68°F–77°F). Protect from light and freezing. After the vial has been punctured, it remains viable for up to 28 days when stored under the same conditions. Always inspect the solution before use — discard if cloudy, discolored, or if the vial seal has been compromised.
Shipping & Handling
Orders placed before 2:00 PM (MST) ship same business day. All vials are sealed, in-date, and shipped in protective packaging to prevent damage in transit. Tracking information is provided via email upon dispatch.
Frequently Asked Questions
Is Hospira bacteriostatic water the same as sterile water?
No. Sterile water contains no preservative and must be used immediately. Bacteriostatic water contains 0.9% benzyl alcohol, which prevents bacterial growth and allows the vial to be reused for up to 28 days after opening.
How long does Hospira bac water last after opening?
Per the manufacturer’s label, bacteriostatic water should be used within 28 days of the initial puncture when stored at controlled room temperature.
Why is Hospira the preferred brand?
Hospira (a Pfizer company) is one of the largest and most trusted U.S. manufacturers of injectable pharmaceuticals. Their bacteriostatic water is produced under FDA-regulated cGMP standards, ensuring consistent quality, sterility, and purity — which is why it is the industry-standard choice for research professionals.
What size is this vial?
This listing is for a single 30mL multi-dose vial (MDV) of Hospira Bacteriostatic Water for Injection, USP.
Do you ship fresh stock?
Yes. All vials are in-date with long expiration windows and shipped directly from a temperature-controlled U.S. facility.
Research Use Only. This product is sold strictly for laboratory research purposes. Not intended for human consumption, diagnostic use, or therapeutic application. Keep out of reach of children. By purchasing, you confirm you are a qualified researcher or professional using this product in accordance with all applicable laws.
{
“@context”: “https://schema.org/”,
“@type”: “Product”,
“name”: “Hospira Bacteriostatic Water for Injection 30mL”,
“image”: [
“https://guardianlabs.life/wp-content/uploads/2026/04/bacteriostaticwater_single.jpg”
],
“description”: “Hospira Bacteriostatic Water 30mL multi-dose vial preserved with 0.9% benzyl alcohol. Pharmaceutical-grade sterile diluent manufactured by Hospira (Pfizer) — the industry-standard choice for peptide reconstitution and laboratory research.”,
“sku”: “HOSPIRA-BACWATER-30ML”,
“mpn”: “0409-4887-17”,
“brand”: {
“@type”: “Brand”,
“name”: “Hospira”
},
“manufacturer”: {
“@type”: “Organization”,
“name”: “Hospira, a Pfizer Company”
},
“category”: “Laboratory Supplies > Diluents”
}















Reviews
There are no reviews yet.